newYou can now listen to Fox News articles!
The U.S. Food and Drug Administration issued a rule on Tuesday making certain types of hearing aids available over-the-counter without a prescription or physical exam.
The FDA said the rule creating a new regulatory category for over-the-counter hearing aids would apply to people “who are perceived to have a mild to moderate hearing impairment.”
“Creating a regulatory category for OTC hearing aids and amending existing regulations will not only provide reasonable assurances of the safety and effectiveness of these devices, but will also promote access and innovation in hearing aid technology. , thereby protecting and promoting public health,” says Rule.
The FDA claims the rule will also reduce the cost of hearing aids. This follows President Biden’s July 2021 Executive Order on Promoting Competition in the U.S. Economy, which clarified that the Secretary of Health and Human Services should issue a “proposed rule for over-the-counter hearing aids.” said to
“It’s the latest action we’re taking to make our economy more competitive and less concentrated,” Biden said in a statement Wednesday. “Too few companies dominate and American consumers pay higher costs. We are finally building an economy that works for working families.”
FDA issues proposal to create new category of hearing aids on the market
Kim M. Smith, Utah deaf hospital rights movement leader and president of the Utah Association for the Deaf, brushes hair off his hearing aids while posing for a portrait at Alta View Hospital Monday, Jan. 20, 2020. I’m here. Sandy, Utah.
(Isaac Hale/The Daily Herald via AP)
The final version of the rule was developed following a notice and comment period that received more than 1,000 public comments from hearing aid manufacturers and consumers, professional groups, legislators and state agencies, the FDA said. . This changed the proposed rules, including lowering the maximum audio output of OTC hearing aids and requiring adjustable volume controls.
The FDA says the rule will go into effect in mid-October, at which point Americans will be able to find over-the-counter hearing aids in drug stores and other retailers, including online.
Democrats claim wins on climate and prescriptions after lawmakers review social tax and spending bills
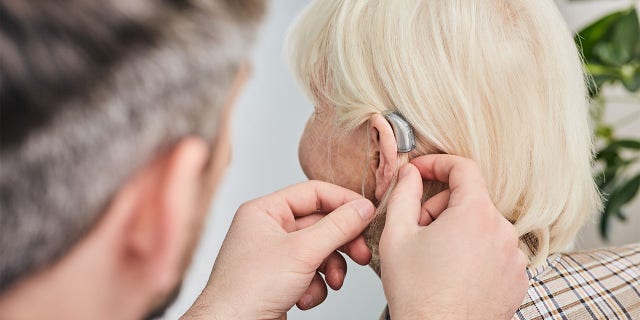
The Food and Drug Administration issued a proposal Tuesday to provide over-the-counter hearing aids.
(iStock)
Health and Human Services Secretary Xavier Becerra said in a statement, “Reducing health care costs in America has been a priority of mine since day one, and this rule will help millions of Americans in need get quality health care.” It is hoped that this will help achieve access to high, affordable healthcare,” said the FDA. It’s an important milestone.”
CLICK HERE TO GET THE FOX NEWS APP
The FDA has specified that the new category of OTC devices “applies to certain air conduction hearing aids intended for persons age 18 and older with perceived mild to moderate hearing impairment.” Others still require a prescription, whether it’s another kind of aid or for younger people.

